|
The Chemical Abstracts Service registry number is a unique identifier of a particular chemical, designed to prevent confusion arising from different languages and naming systems.ĭata for this section been provided by the British Geological Survey.Īn integrated supply risk index from 1 (very low risk) to 10 (very high risk). Where more than one isotope exists, the value given is the abundance weighted average.Ītoms of the same element with different numbers of neutrons. This is approximately the sum of the number of protons and neutrons in the nucleus. 
The mass of an atom relative to that of carbon-12. The transition of a substance directly from the solid to the gas phase without passing through a liquid phase.ĭensity is the mass of a substance that would fill 1 cm 3 at room temperature. The temperature at which the liquid–gas phase change occurs. The temperature at which the solid–liquid phase change occurs. The arrangements of electrons above the last (closed shell) noble gas. These blocks are named for the characteristic spectra they produce: sharp (s), principal (p), diffuse (d), and fundamental (f). The atomic number of each element increases by one, reading from left to right.Įlements are organised into blocks by the orbital type in which the outer electrons are found. 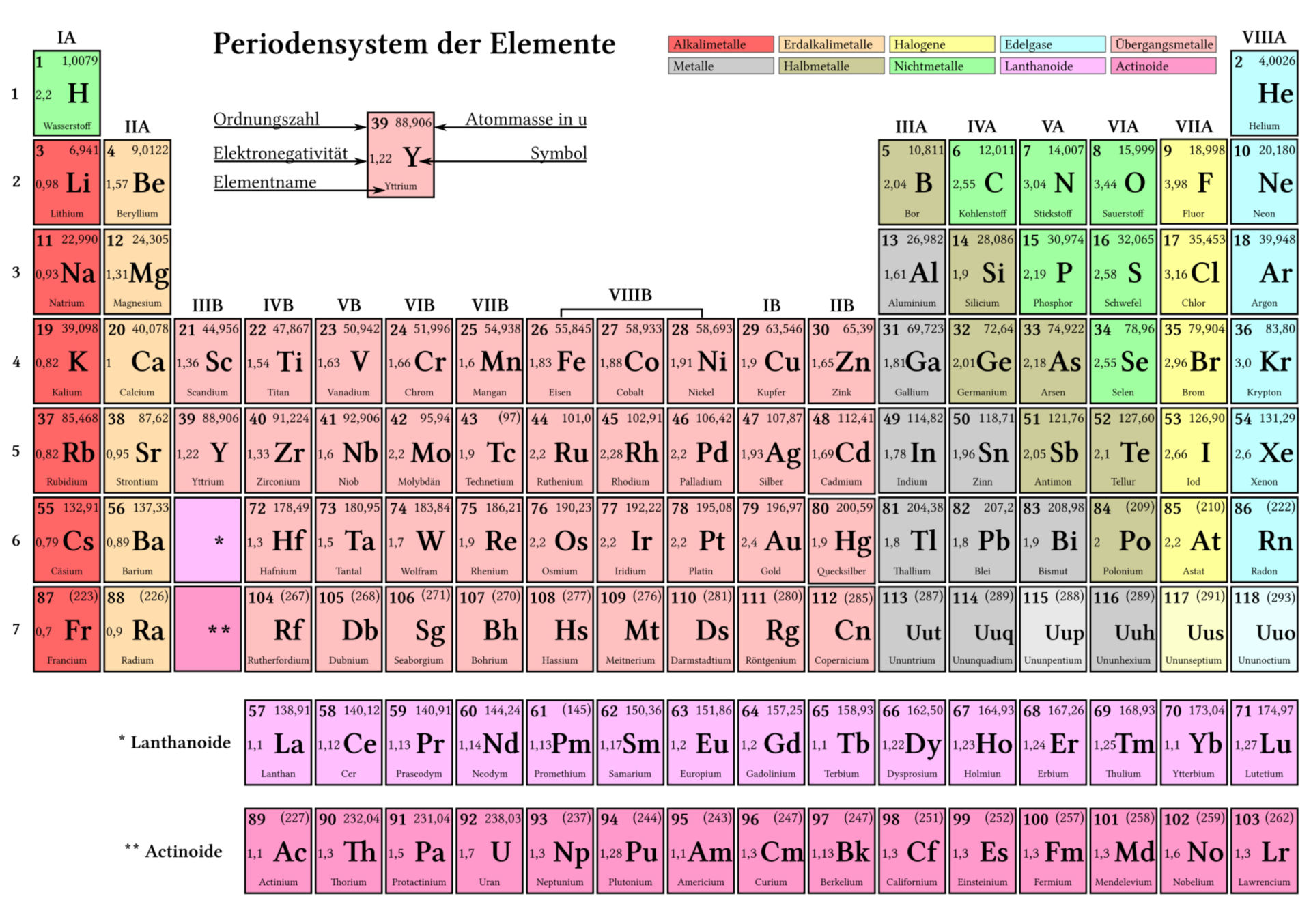
Members of a group typically have similar properties and electron configurations in their outer shell.Ī horizontal row in the periodic table. Trending Questions What Chemical Formula of tuff in chemistry? What jewelry substance contains trapped insects? What are the gases evolved from Na2CO3 plus HCL? What did twiztid member the monoxide child overdose on? What type of molecular force is present in H2? Could you add drops of a solution that was indicated acid into a solution that was indicated base until they became neutral? What is sulphate? How many grams of nitrogen are there in 7.5g of Ca NO3 2? What gases make pink? Where do you find gadolinium? What are three safe methods to test for an acid? What is the empirical formula of a compound that contains 69.24 percent gallium and 30.76 percent phosphorus? What causes fossil fuels to form? How many grams of NaOH are in 1.A vertical column in the periodic table. Calculate the percentage of water in hydrate.Divide the molar mass of water by the molar mass of the hydrate, and multiply result by 100%.36.04g147.01g x 100%Percent water in hydrate is 24.52%. This number may be useful to remember on the day of the test or while doing practice problems.*2. Add these values together to find the molar mass of the hydrate.Molar Mass Anhydrate + Molar Mass Water Molecules* = Molar Mass Hydrate* Tip: the molar mass of water for all hydrate calculations is 18.02g x number of water molecules. Do this for both the anhydrate and the water molecules. 
etc.Add up all the mass values and you have the value for molar mass. Find the molar mass of the hydrate (Calcium Chloride Dihydrate).Find the molar mass of water and the anhydrate (anhydrate + water = hydrate) add the molar mass values of each to find the molar mass of the hydrate.Molar Mass CaCl2: 110.98g+ Molar Mass H2O: 36.04g*Molar Mass CaCl2 * 2H2O: 147.01gFinding Molar Mass# atoms element A * atomic mass element A = Mass A# atoms element B * atomic mass element B = Mass B. What is the theoretical percentage of water of hydration in cacl2-2h20?ġ.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
